Culex
Bioanalytical System Inc.
The Culex Automated Pharmacology System is an instrument system designed by Bioanalytical System Inc. to collect pharmacokinetic and pharmacodynamic data during the course of pharmacokinetics or drug metabolism experiments.
News
11.20.2008
BASi Announces New Culex-L Large Animal Automated In Vivo Sampling System
more>>
Culex-L Large Animal Automated In Vivo Sampling System
Collecting biofluid samples or physiological and behavioral data from animals presents challenges from excessive human intervention, including the stress of manual sampling. To reduce the impact of manual sampling and provide more PK/PD data, BASi (West Lafayette, IN, USA) first developed the Culex system for automated blood sampling in rodents. The Culex system can collect blood samples without human presence or intervention, and at the same time permits the simultaneous monitoring of behavioral and pharmacodynamic parameters for a more complete picture of dose effects.
Since the pig is increasingly recommended in PK/PD and toxicology studies for its similarities to humans, we scaled the technology in the Culex system to the pig. This introduces the controls and mechanics of the Culex-L system, reviews its advantages in reducing stress during blood sampling and other operations, and shows its ability to measure accurately and precisely drug concentrations during pharmacokinetic experiments.
Basic Design
The Physical Barrier

- Octagonal, 1.2 m diameter pen with solid walls, plastic-coated perforated floor, self-contained feed trough and in-built water supply.
- The pig moves relative to the pen but remains stationary relative to the exterior space.
- Clockwise and counter-clockwise rotational capabilities.
- A pig fitted with a harness, connected via an umbilicus to a sensor array above the center of the pen which detects twisting of the umbilicus past pre-established settings, causing the pen to rotate counter to the pig movement.
- Can be used to collect different bio-fluid samples (blood, micro-dialysis and ultra-filtration collections, urine and feces) depending on project need.
- Animal has free access to food and water.
The Harness, Catheter and Blood Lines

- Harness worn by animal over its shoulders and around its chest.
- Secures; a. Blood line attached to catheter(s) implanted in the animal, and, b. Physiological data-generating devices.
- 2 ml – 50 µl draws possible. 24-vial refrigerated storage unit.
- Unused blood in the tubing returned to the animal after collection is complete. Tubing is rinsed with saline after sampling. Intermittent saline pulses given as well to maintain catheter patentcy.
- Monitors to capture physiologic data are either attached to the implanted catheter or secured in the harness.
- Can sample venous or arterial blood.
- Gentle blood sampling procedure does not cause hemolysis.
Automated Control Module for Continuous or Discrete Monitoring

- Software system and blood collection tray for taking and storing blood samples and physiologic data measurements.
- Walk-away capability to draw, collect and store blood samples at whatever time intervals required by project parameters. Can also automatically dose an animal at specific time intervals.
- Catheter is attached for the duration of your study, eliminating interanimal variability (same animal used for longer period of time) in drug testing.
- Capability to track individual physiological parameters and develop correlations between each parameter and contents in the blood. One Pig = multiple data streams.
- Physiologic data include blood pressure, body temperature, respiratory rate, electrocardiogram, and heart rate.
How It Works
The key technology of the Culex system is the clockwise and counter-clockwise rotational capabilities in response to animal movement. A pig placed in the pen is fitted with a harness and connected via an umbilicus to a sensor array which controls the pen movement so the pen rotates counter to any pig movement past pre-established settings. This allows the pig to move relative to the pen but remain stationary relative to the exterior space. The blood collection and drug administration lines are then free of any swivels or unions, from the point of sampling to the delivery of sample into the collector vial. Physiological data leads run from the animal to data collection instruments outside the pen without need for electrical commutator.
Using a prototype system, 4 Gottingen minipigs and 8 commercial feeder pigs (15-30 kg) with surgically implanted jugular catheters were tested for periods of up to 17 days, with 130 or more blood samples drawn with no further handling of the animals. We observed no twisted or blocked tubing due to failure or malfunction of our device. Plasmas from blood samples were clear and non-hemolyzed. We were able to protect tubing connected to a freely-moving pig over an extended period of time, allowing for multiple, high-quality, automated blood samples to be taken without human intervention or physical manipulation of the pigs. This system has been in use now for over a year, and a second prototype design has been finalized.
Evaluating Stress during Blood Sampling
Stress-induced cortisol (CORT) and norepinephrine (NE) were measured as a function of the blood sampling process.
Sixteen pigs (30 kg) were assigned to either (i) automated blood sampling (Culex, BASi) via an indwelling catheter using the PigTurn® penning system, which counter-rotates with the pig’s allowing free movement while preventing catheter twisting; (ii) automated sampling while exposed to visual and auditory responses of manually sampled pigs; (iii) manual sampling by jugular venipuncture. During sampling of group (i), personnel were not permitted in the room; samplings of (ii) and (iii) were performed simultaneously in the same room. Blood samples were collected every 20 min for a total of 120 min and measured for CORT (ng/mL) using mass spectrometry and NE (pg/mL) using HPLC. Effects of treatment and time were computed with mixed models adjusted by Tukey’s post-hoc test.
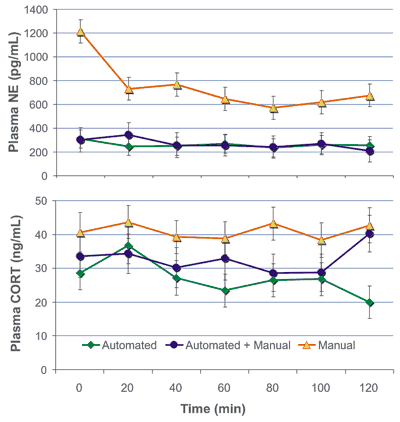

From the data, manual sampling is extremely stressful, but automated sampling, whether or not in the presence of human activities in the vicinity, demonstrated the lowest blood concentrations for these markers.